GD — Society for Dermopharmacy
 |
Issue 1 (2003) |
Authors'
Articles
A. Schrader*, S. Benard and
M. Rohr
The
Rationale and Practice of Biophotonics
* Institute Dr. Schrader - Creachem GmbH, Holzminden, Germany
This
paper was presented at a symposium of the GD Society for Dermopharmacy
in
Duesseldorf, October 17th 2001.
Synopsis
Protecting human skin from the adverse effects of the sun's rays continues
to pose challenges to the pharmaceutical and cosmetic industry. New strategies
for efficacy testing of pharmaceutical and cosmetic formulations are thus
of major interest.
This study was conducted to determine in vivo the UVA protective effect
of chemical filters and antioxidants in complex formulations by means
of ICL-S (Induced Chemiluminescence of Human Skin). This method directly
translates the principles of biophotonics research into practice and allows
the in vivo quantification of stress by measuring the photon emission
of the human skin immediately after exposure to the sun.
UVA radiation (320 - 400 nm) causes an increased formation of free radicals
and reactive oxygen species in the cells. This "oxidative stress"
can damage elementary cellular constituents like DNA, proteins and lipids
and might cause premature skin aging (sun aging) and skin cancer. Part
of the energy involved in these exergonic reactions is released in the
form of photons. This UV-induced but chemically generated photon emission
(chemiluminescence) correlates with the extent of the damage and thus
correlates to a realistic stress inducer (UVA radiation) with biochemical
and biophysical effects on a cellular level.
The technical means of detecting ICL-S involves the use of highly sensitive
photomultipliers and the single photon counting method.
The data generated in volunteer studies show that photon emission is reduced
by up to 70 % in skin treated with a 2 % concentration of chemical UVA
filters compared to untreated skin. The almost 20 % reduction of photoemission
compared to baseline after two weeks of regular application of the product
is evidence of the efficacy of the antioxidants used.
Introduction
Since time immemorial, enzyme catalysed light emission by living organisms (bioluminescence) or the spectacle of shimmering celestial lights such as Aurora borealis (chemiluminescence) has held a magical fascination for man. No less fascinating, however, are the prospects offered by the rational and scientifically correct approach to photon emission phenomena with the aid of high-sensitivity photomultipliers. Biophotonics has even been designated a priority subject in the Recommendations of the Agenda of the German Federal Government "Optical Technologies for the 21st Century".
Ultraviolet radiation and short-wave visible radiation induce a delayed chemiluminescent signal after application to surface areas of living organisms [1, 2]. The nature of such photoinduced emissions is still a focus of research interest. The precise aetiology of the measured chemiluminescence is difficult to determine for a complex organ like the skin. Rather, it is measured as the culminating point of a cascade of reactions characterised by several emission and absorption processes. Influencing the measured signals themselves, however, presents no difficulties.
Especially UVA irradiation causes oxidative stress in cells which severely disturbs the steady state equilibrium (oxidative homeostasis) between oxidative processes and anti-oxidative protective systems [3]. As a result of the increased formation of free radicals and reactive oxygen species (ROS), especially superoxide anion radicals (O2•-) and hydrogen peroxide (H2O2), elementary cellular constituents are altered and their function disturbed. Further reactive species are formed from O2•- and H2O2 together with metal ions, peroxidases or nitrogen monoxide. Hydroxyl radicals (•OH), peroxynitrite, singlet oxygen, hypochloric acid and peroxidase complex I have a particularly marked tissue damaging potential. The absorption of radiation by biologically relevant molecules such as heme compounds or flavines is the primary trigger event. After initiation of these primary oxidative processes, mainly reactions between ROS and unsaturated fatty acids, nucleic acids and proteins are described.
The incidence of chronic radiation damage to the skin has increased dramatically in recent years. Besides acute skin damage, for which the UVB component of sunlight is mainly responsible, UVA radiation (320 - 400 nm) is regarded as the agent causing long-term damage such as premature skin aging (sun aging), immunosuppression and skin cancer [4].
The presence of UVB filters in cosmetic formulations creates a situation in which the skin's early warning signal (sunburn) is activated either not at all or only after prolonged irradiation, thereby further increasing skin exposure to UVA. This makes UVA radiation one of the predominant stress factors for the skin, especially after lengthy exposure to sunlight.
The efficacy of a sun protection product can best be optimised by describing the detrimental factors against which an active substance is intended to protect the skin, as comprehensively as possible in vivo. When in vivo detection of the emitted photons is used as a test method, it can be ensured that a realistic stress inducer (UVA radiation) is linked to a full cellular response, thereby providing the basis for optimised product development.
|
Fig.
1: Causality chain of photon emission
induced by UV radiation.
|
Material and Methods:
Model Formulations
The experiments shown were performed using two different filter systems (UVA1, UVA2) in different concentrations in O/W emulsions. The formulations were prepared by the conventional methods as already described in [5]. In addition to the UVA filters, UVB filters were also included as a reference. The active substances and their concentrations are listed in Tab. 1.
Reference formulations with antioxidants in various combinations and encapsulations were also studied. Tocopheryl acetate was used in a concentration of 3 %, tocopherol (encapsulated) in a concentration of 0.3 %. An antioxidant mix described in literature comprising 1.0 % tocopheryl acetate, 0.25 % rutin and 0.125 % ferulic acid served as control [6].
|
Table.
1:
Reference formulations - chemical UVB and UVA filters used in percent
by weight and their formulation codes. |
ICL-S
(Induced Chemiluminescence of Human Skin)
The device for the measurement of photon emission operates on the pulse counting principle (single photon counting). The light quanta emitted by the skin are detected and amplified by a photomultiplier (PMT) with optimum collection efficiency. A completely new system, which can detect the chemiluminescent signal in vivo immediately after irradiation of the skin, was developed for this purpose (m-u-t GmbH, Wedel, Deutschland). A system of liquid light guides allows defined UV doses to be applied to the skin, followed immediately by photon emission detection.
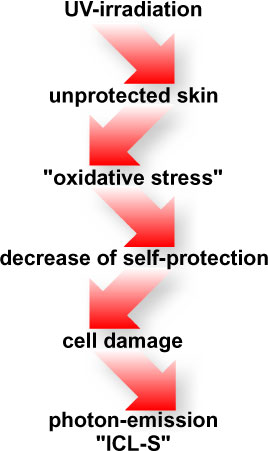
Fig. 2 shows a schematic diagram of the ICL-S system. In order to rule out any type of scattered light, the detector unit and the volunteer remain throughout the measurement in a completely darkened room which meets the high requirements for routine volunteer screening.
Using Peltier elements, the photomultiplier is cooled to a working temperature of about 8 °C to obtain an optimal signal-to-noise ratio and a dark counting rate of less than 10 cps (counts per second). A dome consisting of several optical waveguides with a centrally arranged liquid light guide for irradiation are located at an optimised distance from and angle to the skin without touching the skin. The liquid light guides are contained in the measurement head which can be pressed firmly onto the skin. This arrangement allows a defined photon transfer from the skin to the detector, ensuring maximum reproducibility and compensating factors such as perspiration or slight movements of the volunteers. The combination of optical waveguides and computer-controlled shutters allows the photon emission measurement to be performed immediately after irradiation.
|
Fig.
2:
Experimental arrangement for in vivo measurement of photon emission
on volunteers.
Enlarged version |
Test Design
The studies on the UVA protective efficacy of cosmetic formulations by means of ICL-S were carried out on volunteers of both sexes with healthy skin (n = 20). The lower and middle back served as the test area. In accordance with the COLIPA guidelines for the determination of sun protection factor, 2 mg/cm² of the product were applied to the skin 15 min before each measurement [7, 8]. The test areas had a diameter of 80 mm with a central measuring field of 25 mm diameter. A xenon short arc lamp (m-u-t GmbH, Wedel, Germany) was used as the source of radiation. The continuous spectrum was reduced to the UVA active wavelength of 320 - 400 nm by means of cut-off filters. The radiation dose applied was below 1 J/cm² ( max = 350 nm) and caused no visible changes to the skin.
To determine the ICL-S of the antioxidants used, the test products were applied topically twice daily over a two-week period. The photon emission of the skin was measured before the application phase to document the baseline and about 6 hours after the last product application. The radiation dose was again 1 J/cm² ( λmax = 350 nm).
In addition to the ICL-S investigations, the sun protection factor (SPF) of the study formulations was determined in accordance with COLIPA [7].
Results
UVA Product Performance
To create a validated basis for the ICL-S studies, the sun protection factor of the two O/W formulations, UVB1 and UVB2, was determined according to COLIPA. The sun protection factors for UVB1 and UVB2 were calculated to be SPF = 7 and SPF = 14 respectively and were based purely on the use of chemical UVB filters (see Tab. 1).
In accordance with the test conditions described above, the ICL-S was measured on four test areas after application of the test products which contained exclusively UVB filters (UVB1, UVB2), and those products containing additionally 2 % UVA filters (UVB1 + UVA1, UVB2 + UVA1). An untreated skin area (untreated) served as a control. UVA irradiation of the skin greatly increased the photon emission, which exhibited a typical decay pattern as a function of time (see Fig. 3). Within a few minutes after UVA irradiation, the ICL-S signal of the skin had decreased to a level where no increased photon emission of the skin was measured compared to baseline.
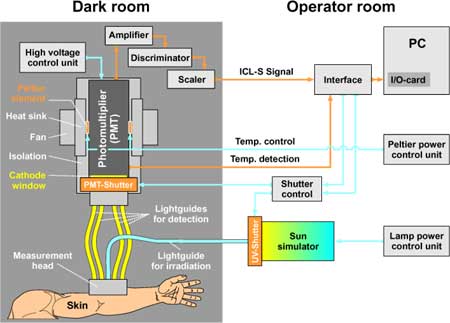
Fig.
3: Typical example of the decay pattern
of the ICL-S. The UVA irradiation ends at time t = 0 s. The grey shaded
area indicates the time period t of the system required to switch
from irradiation to detection.  Enlarged version |
If the integral below these decay functions is used as the basis for further analyses, the untreated skin area shows the highest absolute counting rates. Fig. 4 shows the integral ICL-S of the untreated area and product areas. With pure UVA irradiation, an increase in the UVB filter concentration in the sun protection product applied to the skin does not produce a significant change in the measured integral ICL S. The admixture of 2 % UVA1, however, significantly reduces the measured signal intensity for the formulation with a high and with a low SPF to an equal extent.
Fig. 4A illustrates this situation with the mean integral counting rate of 20 volunteers. The integral calculations were based on the area under the decay functions within a time interval of 1-200 s after UVA irradiation. The integral counting rates for the untreated area and the pure UVB protection of the skin are approximately 25000 integral counts (cps s). The additional use of UVA1 results in measured signals below 10000 cps s. Fig. 4B presents the results as a percentage decrease with reference to the untreated test area. An insignificant reduction of less than 5 % is observed for the UVB filter combinations UVB1 and UVB2, while after addition of 2 % UVA1 significant reductions of 64 % and 73 % result for the combinations of UVB1 and UVB2 with UVA1.
| Fig. 4: Mean
integral ICL-S (n = 20) immediately after UVA irradiation. The absolute integral signal intensity (A) and the data normalised to the untreated area (B) are shown. For details of the active substances used see Tab. 1 ( p 0.05).  Enlarged version |
When the integral counting rates of the untreated area are expressed in relation to the product areas, size-independent ratios of 3 and 2 are obtained for the UVB1 and UVB2 formulations respectively, and a value of 14 for the UVB1+UVA1 and UVB2+UVA1 formulations. The values for the skin area treated with placebo are within the same range (about 2.5) as the values measured for the skin areas treated with UVB filter. The in vivo results demonstrate that the UVA protection afforded by a product can be determined by means of ICL-S irrespective of the existing UVB protective properties.
To verify this result, serial concentrations of a further filter with good UVA-absorbent properties were tested. 0.5, 1.0 and 2.0 % UVA2 were used on three test areas. Chemical UVB filters were added to these formulations on three further test areas (see Tab. 1). The influence of the base itself on the signal intensities was tested using a placebo containing no filter.
Fig. 5 shows the decay kinetics immediately after UVA irradiation of the skin in a double logarithmic reference frame. The untreated measured area is seen to have the highest signal intensities. Furthermore, a continuous decrease in the signal intensities was observed as the UVA filter concentration increased. The measured ICL-S signal had already regained its initial level 100 s after application of the harmful UV radiation.
| Fig.
5: Mean decay kinetics (n = 20) immediately after UVA irradiation.
For details of the active substances used see Tab. 1. 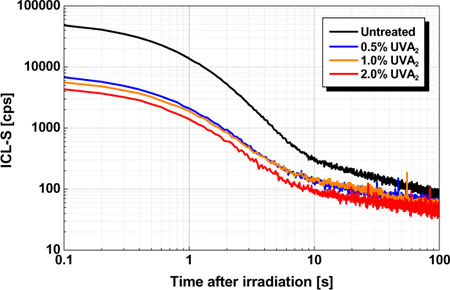
Enlarged version |
Normalisation of the values to untreated area and placebo reveals a decrease in the integral ICL-S of about 60 % regardless of the fact that the reference product contains an additional UVB protection of SPF 7 (see Fig. 6).
| Fig.
6: Correlation of the measured ICL-S normalised to the untreated
area with the UVA filter concentration used (n = 20). For details of the active substances used see Tab. 1. 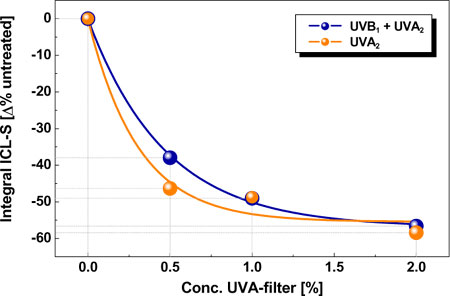
Enlarged version |
Based on these data, ratios of about 7 were obtained for a UVA filter concentration of 0.5 %, and 8 for a filter concentration of 1 %. When using 2 % of the new UVA filter, a value of about 10 was obtained when the measured integral counts of the untreated area were expressed in relation to those of the product areas. Fitting these data reveals a logarithmic dependence on the calculated ratios on the UVA filter concentration used.
Antioxidative Potential
of Active Ingredients
Besides these "primary" UVA protection products, which provide skin protection by shielding the skin from the harmful environmental agent, secondary cellular protection by increasing the skin's endogenous antioxidative potential is also conceivable. With this approach the sun's radiation is not kept away from the skin, instead the formation of free radicals and ROS induced by UVA radiation is reduced and their effects on cellular components diminished.
Fig. 7 shows the reduction of the measured integral photon emission by topically applied antioxidants normalised to the respective baseline value. The initial situation of the skin was documented by determining the ICL-S before product application. The volunteers were then treated with the different test products with instructions to apply them twice daily over a two-week period. The ICL-S was then measured again and these final values were compared to baseline.
Fig.
7: Integral ICL-S after application of the product for 14 days,
normalised to the untreated baseline value ( p 0.05).
Enlarged version |
Use of the base without antioxidant active substances causes no significant change in the photon emission measured and thus also no change in the skin properties or equipment parameters. The formulation with added -tocopherol acetate (Vit-E AC) behaves in a similar way to the placebo. If pure -tocopherol (Vit-E OH) is used, which is additionally encapsulated to prevent autooxidation and allow optimised cellular supply. A reduction in the photon emission of about 7 % is measured compared to baseline. A combination of different antioxidants (AO mix) described in the literature [6], which resulted in a significant reduction of about 17 % compared to the untreated baseline value, was used as a control.
Discussion and Outlook
The ICL-S results presented were generated without exception under in vivo conditions on volunteers and show that topically applied chemical UVA filters can have a significant influence on UVA induced photon emission in the human skin in vivo. Sun protection products formulated exclusively on the basis of chemical UVB filters do not exhibit a concentration dependent correlation with the measured ICL-S on application of pure UVA radiation. Similarly, no correlation is apparent between the measured signal intensities and the previously determined SPF of the products.
These results encourage to pursue further studies in this field, especially since there is still a need for further in vivo determinations of UVA protective efficacy on volunteers. The method used here is suitable for measuring very weak cellular reactions, which makes it possible to drastically reduce the energy used, so that there are no visible changes to the skin. After a few minutes, the induced photon emission itself is no longer distinguishable from the signal of the untreated skin. An exact assignment in vivo of the radical species contributing to the photon emission is not possible at present. Similarly, no conclusive statements can be made regarding the total quantity of photons induced in the cells. A quantification of the emitted photons which are detected as endpoint in a complex chain of reactions which start with UVA irradiation is practicable.
Further studies in the concentration range below 0.5 % of the UVA filters used are planned, with the aim of describing the relationship between in vivo protective efficacy and measured ICL-S signal intensity with even greater mathematical accuracy. Also planned are comparative measurements with existing methods which also quantify the protective efficacy of cosmetic formulations.
Antioxidant-based cellular protection produces a much smaller reduction in the measured ICL-S than that achieved by primary UVA protection with topically applied filter substances. Sufficient protection of the skin against UVA radiation can therefore not be achieved with antioxidants alone. Indeed, it appears that a combination of UVA filters and antioxidant agents aimed at optimising product properties does make sense. When performing in vivo measurements by means of ICL-S, the primary concern is to ensure that the substances used actually reach their site of action. Many in vitro methods for determining the antioxidant potential of active ingredients do not take this fact into account.
Some antioxidant substances have a distinct molecular structure that allows them to interact directly with UVA radiation. With a suitable test design and using a low concentration of active ingredients of the antioxidants, however, their UVA filter properties on the skin are of secondary importance.
Molecules that are chemiluminescent under certain conditions have been used routinely for a long time as sensitive markers, especially in medical and biochemical diagnostics. In the pharmaceutical and cosmetics field, in vivo measurements on volunteers are not yet routine practice because of the high equipment costs involved, but the results presented demonstrate the high potential of biophotonics for use in pharmaceutical and cosmetic research. Besides in vivo measurements on volunteers, other areas within cosmetics might well also require non-invasive testing, as recent research has revealed [9].
Acknowledgement
The authors would like to thank Prof. Dr. Heering, Phototechnical Institute - Karlsruhe University, and the m-u-t GmbH company for their many years of good cooperation and the technical realisation of the measuring devices and the Bundesministerium für Wirtschaft, the Arbeitsgemeinschaft industrieller Forschungsvereinigungen e.V. (AiF), the Forschungsgemeinschaft für die kosmetische Industrie e.V. (FKI). Part of this work was supported by the AiF (Project No. 11881N). This research project is funded from the budget of the German Federal Ministry of Economics through the non-profit Task Force of Industrial Research
Associations "Otto von Guericke" e.V. (AiF).
References
[1] Niggli, H.J. (1993) Artificial sunlight irradiation induces ultraweak photon emission in human skin fibroblasts. J. Photochem. Photobiol. B., 18, 281-285.
[2] Sauermann, G., Mei, W.P., Hoppe, U., Stäb, F. (1999) Ultraweak photon emission of human skin in vivo: influence of topically applied antioxidants on human skin. Methods Enzymol., 300:419-28, 419-428.
[3] Podda, M., Traber, M.G., Weber, C., Yan, L.J., Packer, L. (1998) UV-irradiation depletes antioxidants and causes oxidative damage in a model of human skin. Free Radic. Biol. Med., 24, 55-65.
[4] Krutmann, J. (2000) Photoaging, photodermatoses, and photocarcinogenesis: recent advances in understanding the detrimental effects of ultraviolet-A radiation on human skin. IFSCC magazine, 3, 51-53.
[5] Schrader, K. (1990) Die Entwicklung und Prüfung von Lichtschutzpräparaten. SÖFW Journal, 10, 400-407.
[6] Stäb, F., Mundt, C., Blatt, T., Will, K., Keyhani, R., Rippke, F., Max, H., Schönrock, U., Wenck, H., Moll, I., Hölzle, E., Wittern, K.-P. (2000) Alpha-glucosylrutin (AGR) - an innovative antioxidant in skin protection. IFSCC magazine, 3, 39-43.
[7] Rohr, M., Schrader, K. (1998) Climatic influence on cosmetic skin parameters. Corr. Probl. Dermatol., 26, 151-164.
[8] Rohr, M., Schrader, A., Schrader, K. (1998) Die Bestimmung des Sonnenschutzfaktors nach COLIPA. Parfümerie und Kosmetik, 5, 12-19.
[9] Benard, S., Nerenz, H., Rohr, M., Schrader, K. (2001) ICL-H (Induced chemiluminescence of human hair) - a new method for quantitative analysis of hair stress. IFSCC magazine, 4, 185-189.
top